 W
WO-Acetylpsilocin is a semi-synthetic psychoactive drug that has been suggested by David Nichols to be a potentially useful alternative to psilocybin] for pharmacological studies, as they are both believed to be prodrugs of psilocin. [. However, some users report that O-acetylpsilocin's subjective effects differ from those of psilocybin and psilocin. It is the acetylated form of the psilocybin mushroom alkaloid psilocin and is a lower homolog of 4-AcO-MET, 4-AcO-DET, 4-AcO-MiPT and 4-AcO-DiPT.
 W
WAeruginascin or N,N,N-trimethyl-4-phosphoryloxytryptamine is an indoleamine derivative which occurs naturally within the mushroom Inocybe aeruginascens and Pholiotina cyanopus. Aeruginascin is the N-trimethyl analogue of psilocybin. It is closely related to the frog skin toxin bufotenidine (5-HTQ), a potent 5-HT3 receptor agonist. The only scientific literature about the pharmacological effects of aeruginascin is from a study published by Gartz in 1989. Across 23 analyzed cases of accidental hallucinogenic mushroom poisonings, people who had ingested the mushroom Inocybe aeruginascens reported only euphoric experiences. This is in contrast to the slight and in some cases extremely dysphoric experiences reported from the accidental ingestion of non aeruginascin containing mushrooms(containing solely psilocybin and psilocin).
 W
WAgaritine (AGT) is an aromatic, antiviral, hydrazine-derivative mycotoxin and IARC Group 3 carcinogen that occurs in mushroom species of the genus Agaricus.
 W
WAlternariol is a toxic metabolite of Alternaria fungi. It is an important contaminant in cereals and fruits. Alternariol exhibits antifungal and phytotoxic activity. It is reported to inhibit cholinesterase enzymes. It is also a mycoestrogen.
 W
WAristolochene is a bicyclic sesquiterpene produced by certain fungi including the cheese mold Penicillium roqueforti. It is biosynthesized from farnesyl pyrophosphate by aristolochene synthase and is the parent hydrocarbon of a large variety of fungal toxins.
 W
WAsperlicin is a mycotoxin, derived from the fungus Aspergillus alliaceus. It acts as a selective antagonist for the cholecystokinin receptor CCKA, and has been used as a lead compound for the development of a number of novel CCKA antagonists with potential clinical applications.
 W
WAszonalenin is an alkaloid which is produced by Neosartorya and Aspergillus species. Aszonalenin is a neurotoxin.
 W
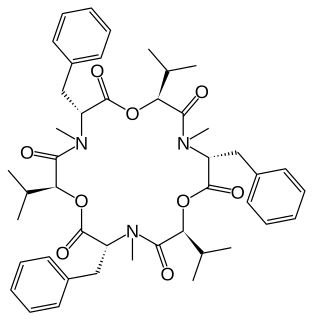
WBeauvericin is a depsipeptide with antibiotic and insecticidal effects belonging to the enniatin family. It was isolated from the fungus Beauveria bassiana, but is also produced by several other fungi, including several Fusarium species; it may therefore occur in grain contaminated with these fungi. Beauvericin is active against Gram-positive bacteria and mycobacteria, and is also capable of inducing programmed cell death in mammals.
 W
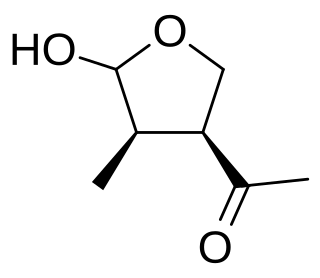
WBotryodiplodin is an antibiotic mycotoxin produced by Penicillium.
 W
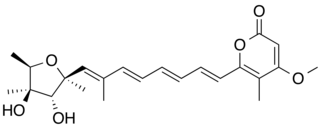
WCitreoviridin is a mycotoxin which is produced by Penicillium and Aspergillus species. If rice, corn, cereals or meat products are contaminated with Penicillin citreoviridin, citreoviridin can be produced if the food is stored in a damp place. Consuming food which is contaminated with citreoviridin can cause the disease cardiac beri beri. Furthermore it damages liver and kidneys.
 W
WCitrinin is a mycotoxin which is often found in food. It is a secondary metabolite produced by fungi that contaminate long-stored food and it causes different toxic effects, like nephrotoxic, hepatotoxic and cytotoxic effects. Citrinin is mainly found in stored grains, but sometimes also in fruits and other plant products.
 W
WCoprine is a mycotoxin. It was first isolated from common inkcap. It occurs in mushrooms in the genera Coprinopsis, with the most popular example being the brawny bolete. When combined with the consumption of alcohol, it causes "Coprinus syndrome". It inhibits the enzyme acetaldehyde dehydrogenase, which is involved in the metabolism of alcohol. This inhibition leads to a buildup of acetaldehyde, causing an alcohol flush reaction. Because of this, the mushroom is commonly referred to as Tippler's Bane.
 W
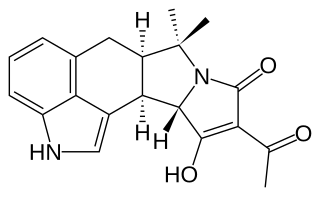
WCyclochlorotine (CC), also known as islanditoxin is a mycotoxin produced by the fungus Penicillium islandicum that causes liver damage and has carcinogenic properties. Japanese researchers confirmed that it was one of three strains of Penicillin fungi responsible for yellowed rice. It is listed as an IARC Group 3 carcinogen.
 W
WCyclopiazonic acid (CPA) is a toxic fungal secondary metabolite. Chemically, CPA is related to ergoline alkaloids. CPA was originally isolated from Penicillium cyclopium and subsequently from other fungi including Penicillium griseofulvum, Penicillium camemberti, Penicillium commune, Aspergillus flavus, and Aspergillus versicolor. CPA only appears to be toxic in high concentrations. Ingestion of CPA causes one to be an anorexic, to be dehydrated, to lose weight, to be immobile, and has some sign of spasm when near death. They can be found in molds, corns, peanuts, and other fermented products, such as cheese and sausages. Biologically, CPA is a specific inhibitor of SERCA ATPase in intracellular Ca2+ storage sites. CPA inhibits SERCA ATPase by keeping it in one specific confirmation, thus, preventing it from forming another. CPA also binds to SERCA ATPase at the same site as another inhibitor, thapsigargin (TG). In this way, CPA lowers the ability of SERCA ATPase to bind an ATP molecule.
 W
WCycloprop-2-ene carboxylic acid is a mycotoxin found in some mushrooms such as Russula subnigricans.
 W
WCytochalasin B, the name of which comes from the Greek cytos (cell) and chalasis (relaxation), is a cell-permeable mycotoxin. It was found that substoichimetric concentrations of cytochalasin B (CB) strongly inhibit network formation by actin filaments. Due to this, it is often used in cytological research. It inhibits cytoplasmic division by blocking the formation of contractile microfilaments. It inhibits cell movement and induces nuclear extrusion. Cytochalasin B shortens actin filaments by blocking monomer addition at the fast-growing end of polymers. Cytochalasin B inhibits glucose transport and platelet aggregation. It blocks adenosine-induced apoptotic body formation without affecting activation of endogenous ADP-ribosylation in leukemia HL-60 cells. It is also used in cloning through nuclear transfer. Here enucleated recipient cells are treated with cytochalasin B. Cytochalasin B makes the cytoplasm of the oocytes more fluid and makes it possible to aspirate the nuclear genome of the oocyte within a small vesicle of plasma membrane into a micro-needle. Thereby, the oocyte genome is removed from the oocyte, while preventing rupture of the plasma membrane.
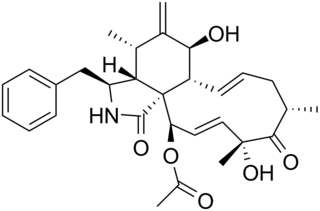 W
WCytochalasin D is a member of the class of mycotoxins known as cytochalasins. Cytochalasin D is an alkaloid produced by Helminthosporium and other molds.
 W
WCytochalasin E, a member of the cytochalasin group, is an inhibitor of actin polymerization in blood platelets. It inhibits angiogenesis and tumor growth. Unlike cytochalasin A and cytochalasin B, it does not inhibit glucose transport.
 W
WFumonisin B1 is the most prevalent member of a family of toxins, known as fumonisins, produced by several species of Fusarium molds, such as Fusarium verticillioides, which occur mainly in maize (corn), wheat and other cereals. Fumonisin B1 contamination of maize has been reported worldwide at mg/kg levels. Human exposure occurs at levels of micrograms to milligrams per day and is greatest in regions where maize products are the dietary staple.
 W
WFumonisin B2 is a fumonisin mycotoxin produced by the fungi Fusarium verticillioides and Aspergillus niger.
 W
WFumonisin B4 is a fumonisin mycotoxin produced mainly by the fungi Fusarium proliferatum, Fusarium verticillioides. Recently FB4 has been detected in fungi Aspergillus niger and in several Tolypocladium species.
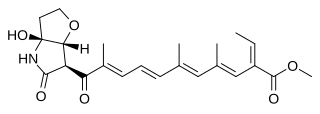 W
WFusarins are a class of mycotoxins produced mainly by fungi of the genus Fusarium, which can infect agriculturally important crops such as wheat, barley, oats, rye, and corn. Chemically, they are polyketides that are also derived from amino acids.
 W
WGliotoxin is a sulfur-containing mycotoxin that belongs to a class of naturally occurring 2,5-diketopiperazines produced by several species of fungi, especially those of marine origin. It is the most prominent member of the epipolythiopiperazines, a large class of natural products featuring a diketopiperazine with di- or polysulfide linkage. These highly bioactive compounds have been the subject of numerous studies aimed at new therapeutics. Gliotoxin was originally isolated from Gliocladium fimbriatum, and was named accordingly. It is an epipolythiodioxopiperazine metabolite.
 W
WA loline alkaloid is a member of the 1-aminopyrrolizidines, which are bioactive natural products with several distinct biological and chemical features. The lolines are insecticidal and insect-deterrent compounds that are produced in grasses infected by endophytic fungal symbionts of the genus Epichloë. Lolines increase resistance of endophyte-infected grasses to insect herbivores, and may also protect the infected plants from environmental stresses such as drought and spatial competition. They are alkaloids, organic compounds containing basic nitrogen atoms. The basic chemical structure of the lolines comprises a saturated pyrrolizidine ring, a primary amine at the C-1 carbon, and an internal ether bridge—a hallmark feature of the lolines, which is uncommon in organic compounds—joining two distant ring carbons. Different substituents at the C-1 amine, such as methyl, formyl, and acetyl groups, yield loline species that have variable bioactivity against insects. Besides endophyte–grass symbiota, loline alkaloids have also been identified in some other plant species; namely, Adenocarpus species and Argyreia mollis.
 W
WMoniliformin is an unusual mycotoxin, a feed contaminant that is lethal to fowl, especially ducklings.
 W
WMonocerin is a dihydroisocoumarin and a polyketide metabolite that originates from various fungal species. It has been shown to display antifungal, plant pathogenic, and insecticidal characteristics. Monocerin has been isolated from Dreschlera monoceras, D. ravenelii, Exserohilum turcicum, and Fusarium larvarum.
 W
WMT81 is a mycotoxin with antibiotic activity.
 W
WMuscarine, L-(+)-muscarine, or muscarin is a natural product found in certain mushrooms, particularly in Inocybe and Clitocybe species, such as the deadly C. dealbata. Mushrooms in the genera Entoloma and Mycena have also been found to contain levels of muscarine which can be dangerous if ingested. Muscarine has been found in harmless trace amounts in Boletus, Hygrocybe, Lactarius and Russula. Trace concentrations of muscarine are also found in Amanita muscaria, though the pharmacologically more relevant compound from this mushroom is the Z-drug-like alkaloid muscimol. A. muscaria fruitbodies contain a variable dose of muscarine, usually around 0.0003% fresh weight. This is very low and toxicity symptoms occur very rarely. Inocybe and Clitocybe contain muscarine concentrations up to 1.6%.
 W
WMuscazone is a toxic chemical compound. It is an amino acid found in European fly agaric mushrooms.
 W
WMuscimol is one of the principal psychoactive constituents of Amanita muscaria and related species of mushroom. Muscimol is a potent, selective agonist for the GABAA receptors and displays sedative-hypnotic, depressant and hallucinogenic psychoactivity. This colorless or white solid is classified as an isoxazole.
 W
WMushroom poisoning refers to harmful effects from ingestion of toxic substances present in a mushroom. These symptoms can vary from slight gastrointestinal discomfort to death in about 10 days. The toxins present are secondary metabolites produced by the fungus. Mushroom poisoning is usually the result of ingestion of wild mushrooms after misidentification of a toxic mushroom as an edible species. The most common reason for this misidentification is close resemblance in terms of colour and general morphology of the toxic mushrooms species with edible species. To prevent mushroom poisoning, mushroom gatherers familiarize themselves with the mushrooms they intend to collect, as well as with any similar-looking toxic species. The safety of eating wild mushrooms may depend on methods of preparation for cooking.
 W
Wbeta-Nitropropionic acid is a mycotoxin, a potent mitochondrial inhibitor, toxic to humans. It is produced by a number of fungi, and found widely in food, in sugar cane, as well as Japanese fungally fermented staples miso, soy sauce, katsuobushi, and some traditional Chinese medicines.
 W
WNivalenol (NIV) is a mycotoxin of the trichothecene group. In nature it is mainly found in fungi of the Fusarium species. The Fusarium species belongs to the most prevalent mycotoxin producing fungi in the temperate regions of the northern hemisphere, therefore making them a considerable risk for the food crop production industry.
 W
WOchratoxins are a group of mycotoxins produced by some Aspergillus species and some Penicillium species, especially P. verrucosum. Ochratoxin A is the most prevalent and relevant fungal toxin of this group, while ochratoxins B and C are of lesser importance.
 W
WOchratoxin A—a toxin produced by different Aspergillus and Penicillium species — is one of the most-abundant food-contaminating mycotoxins. It is also a frequent contaminant of water-damaged houses and of heating ducts. Human exposure can occur through consumption of contaminated food products, particularly contaminated grain and pork products, as well as coffee, wine grapes, and dried grapes. The toxin has been found in the tissues and organs of animals, including human blood and breast milk. Ochratoxin A, like most toxic substances, has large species- and sex-specific toxicological differences.
 W
WPenicillic acid is a mycotoxin that is produced by Aspergillus flavus and Penicillium roqueforti mold. It is also the major product of acid degradation of penicillin. Its first practical synthesis was reported in 1947 by Ralph Raphael, who had worked on penicillin during World War II.
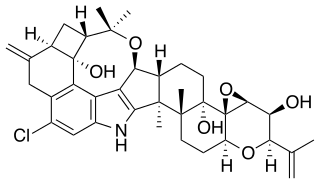 W
WPenitrem A (tremortin) is an indole-diterpenoid mycotoxin produced by certain species of Aspergillus, Claviceps, and Penicillium, which can be found growing on various plant species such as ryegrass. Penitrem A is one of many secondary metabolites following the synthesis of paxilline in Penicillium crostosum. Penitrem A poisoning in humans and animals usually occurs through the consumption of contaminated foods by mycotoxin-producing species, which is then distributed through the body by the bloodstream. It bypasses the blood-brain barrier to exert its toxicological effects on the central nervous system. In humans, penitrem A poisoning has been associated with severe tremors, hyperthermia, nausea/vomiting, diplopia, and bloody diarrhea. In animals, symptoms of penitrem A poisoning has been associated with symptoms ranging from tremors, seizures, and hyperthermia to ataxia and nystagmus.
 W
WThe phomoxanthones are a loosely defined class of natural products. The two founding members of this class are phomoxanthone A and phomoxanthone B. Other compounds were later also classified as phomoxanthones, although a unifying nomenclature has not yet been established. The structure of all phomoxanthones is derived from a dimer of two covalently linked tetrahydroxanthones, and they differ mainly in the position of this link as well as in the acetylation status of their hydroxy groups. The phomoxanthones are structurally closely related to other tetrahydroxanthone dimers such as the secalonic acids and the eumitrins. While most phomoxanthones were discovered in fungi of the genus Phomopsis, most notably in the species Phomopsis longicolla, some have also been found in Penicillium sp.
 W
WThe mycotoxin phomoxanthone A, or PXA for short, is a toxic natural product that affects the mitochondria. It is the most toxic and the best studied of the naturally occurring phomoxanthones. PXA has recently been shown to induce rapid, non-canonical mitochondrial fission by causing the mitochondrial matrix to fragment while the outer mitochondrial membrane can remain intact. This process was shown to be independent from the mitochondrial fission and fusion regulators DRP1 and OPA1.
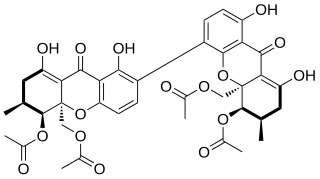 W
WThe mycotoxin phomoxanthone B, or PXB for short, is a toxic natural product. It is a less toxic isomer of phomoxanthone A and one of the two founding members of the class of phomoxanthone compounds. The phomoxanthones are named after the fungus Phomopsis, from which they were first isolated, and after their xanthonoid structure. Chemically, they are dimers of two tetrahydroxanthones that are covalently linked to each other. PXB itself is a homodimer of two identical diacetylated tetrahydroxanthones. The position of the link between the two tetrahydroxanthones is the only structural difference between PXB and its isomers PXA and dicerandrol C: In PXA, the two xanthonoid monomers are symmetrically linked at C-4,4’, while in PXB, they are asymmetrically linked at C-2,4’, and in dicerandrol C, they are symmetrically linked at C-2,2’.
 W
WPenicillin Roquefort Toxin is a mycotoxin produced by the fungi Penicillium roqueforti. In 1973, PR toxin was first partially characterized by isolating moldy corn on which the fungi had grown. Although its lethal dose was determined shortly after the isolation of the chemical, details of its toxic effects, were not fully clarified until 1982 in a study with mice, rats, anesthetized cats and preparations of isolated rat auricle.
 W
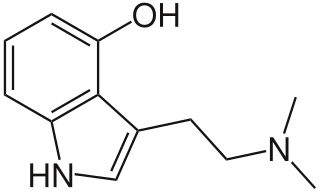
WPsilocin is a substituted tryptamine alkaloid and a serotonergic psychedelic substance. It is present in most psychedelic mushrooms together with its phosphorylated counterpart psilocybin. Psilocin is a Schedule I drug under the Convention on Psychotropic Substances. The mind-altering effects of psilocin are highly variable, subjective and resemble those of LSD and DMT.
 W
WPsilocybin is a naturally occurring psychedelic prodrug compound produced by more than 200 species of fungus. The most potent are members of the genus Psilocybe, such as P. azurescens, P. semilanceata, and P. cyanescens, but psilocybin has also been isolated from about a dozen other genera. As a prodrug, psilocybin is quickly converted by the body to psilocin, which has mind-altering effects similar, in some aspects, to those of LSD, mescaline, and DMT. In general, the effects include euphoria, visual and mental hallucinations, changes in perception, a distorted sense of time, and spiritual experiences, and can also include possible adverse reactions such as nausea and panic attacks.
 W
WPyrenocines are antibiotic mycotoxins.
 W
WRoquefortine C is a mycotoxin that belongs to a class of naturally occurring 2,5-diketopiperazines produced by various fungi, particularly species from the genus Penicillium. It was first isolated from a strain of Penicillium roqueforti, a species commercially used as a source of proteolytic and lipolytic enzymes during maturation of the blue-veined cheeses, Roquefort, Danish Blue, Stilton and Gorgonzola.
 W
WRubratoxins are hepatotoxic mycotoxin produced by Penicillium rubrum and Penicillium purpurogenum. Rubratoxin A and rubratoxin B have been known since 1950s, while rubratoxin C was recently reported. Rubratoxins are also known as protein phosphatase 2A (PP2A) specific inhibitor. The PP2A inhibitory activity of rubratoxin A is about 100-fold higher than rubratoxin B and rubratoxin A is now used as a chemical probe for PP2A research.
 W
WSecalonic acids are a group of chiral dimeric tetrahydroxanthones closely related to ergoflavin and ergochrysin A that are collectively called ergochromes and belong to a class of mycotoxins initially isolated as major ergot pigments from the fungi Claviceps purpurea that grows parasitically on rye grasses. From early times and particularly in medieval Europe the consumption of grains containing ergot has repeatedly lead to mass poisonings known as ergotism which was caused by toxic ergot alkaloids and mycotoxins such as the ergochromes, due to contamination of flour by C. purpurea. A cluster of genes responsible for the synthesis of secalonic acids in C. purpurea has been identified. Secalonic acid D the enantiomer of secalonic acid A is a major environmental toxin, isolated from the fungus Penicillium oxalicum, and is a major microbial contaminant of freshly-harvested corn which causes toxicity through contamination of foodstuffs.
 W
WSlaframine is an indolizidine alkaloidal mycotoxin that generally causes salivation (slobbers) in most animals. It is usually produced by the fungus Rhizoctonia leguminicola. It is a common fungal pathogen of red clover (Trifolium pratense) that causes black patch disease in the plant. Slaframine has the molecular formula C10H18N2O2. The different environmental condition promotes the growth of the Rhizoctonia leguminicola fungus. For example, wet and humid weather are the favorable environmental condition for the growth of the fungus and production of slaframine. Legume hays contaminated with slaframine causes slobber syndrome and the various animals are sensitive to its effects.
 W
WSterigmatocystin is a polyketide mycotoxin produced by certain species of Aspergillus. The toxin is naturally found in some cheeses.
 W
WSterpuric acid is a phytotoxic metabolite derived from the fungus Stereum purpureum, from which it derives its name. This fungus causes silver-leaf disease of fruit trees.
 W
WTaleranol, or teranol, also known as β-zearalanol, is a synthetic, nonsteroidal estrogen of the resorcylic acid lactone group related to mycoestrogens found in Fusarium spp which was never marketed. It is the β epimer of zeranol (α-zearalanol) and is a major metabolite of zeranol but with less biological activity.
 W
WTenuazonic acid is a mycotoxin produced by Alternaria species. It is a powerful eukaryotic protein synthesis inhibitor.
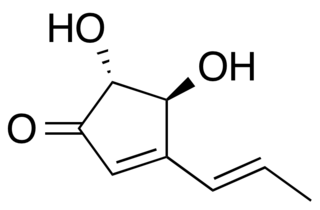 W
WTerrein is a fungal metabolite of Aspergillus species. Terrein forms pale yellow crystal needles. Terrein has a strong cytotoxic activity against cells with colorectal carcinoma. The strain S020 from the fungus Aspergillus terreus has the highest rate in producing terrein.
 W
WVerruculogen is a mycotoxin produced by certain strains of aspergillus that belongs to a class of naturally occurring 2,5-diketopiperazines. It is an annulated analogue of cyclo(L-Trp-L-Pro) which belongs to the most abundant and structurally diverse class of tryptophan-proline 2,5-diketopiperazine natural products. It produces tremors in mice due to its neurotoxic properties. It also tested positive in a Salmonella/mammalian microsome assay and was shown to be genotoxic. It is a potent blocker of calcium-activated potassium channels.
 W
WVomitoxin, also known as deoxynivalenol (DON), is a type B trichothecene, an epoxy-sesquiterpenoid. This mycotoxin occurs predominantly in grains such as wheat, barley, oats, rye, and corn, and less often in rice, sorghum, and triticale. The occurrence of deoxynivalenol is associated primarily with Fusarium graminearum and F. culmorum, both of which are important plant pathogens which cause fusarium head blight in wheat and gibberella or fusarium ear blight in corn. The incidence of fusarium head blight is strongly associated with moisture at the time of flowering (anthesis), and the timing of rainfall, rather than the amount, is the most critical factor. However, increased amount of moisture towards harvest time has been associated with lower amount of vomitoxin in wheat grain due to leaching of toxins. Furthermore, deoxynivalenol contents are significantly affected by the susceptibility of cultivars towards Fusarium species, previous crop, tillage practices, and fungicide use. It occurs abundantly in grains in Norway due to heavy rainfall.
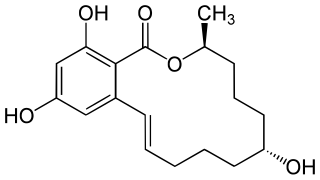 W
Wα-Zearalenol is a nonsteroidal estrogen of the resorcylic acid lactone group related to mycoestrogens found in Fusarium spp. It is the α epimer of β-zearalenol and along with β-zearalenol is a major metabolite of zearalenone formed mainly in the liver but also to a lesser extent in the intestines during first-pass metabolism. A relatively low proportion of β-zearalenol is formed from zearalenone compared to α-zearalenol in humans. α-Zearalenol is about 3- to 4-fold more potent as an estrogen relative to zearalenone.
 W
Wβ-Zearalenol is a nonsteroidal estrogen of the resorcylic acid lactone group related to mycoestrogens found in Fusarium spp. It is the β epimer of α-zearalenol and along with α-zearalenol is a major metabolite of zearalenone formed mainly in the liver but also to a lesser extent in the intestines during first-pass metabolism. A relatively high proportion of α-zearalenol is formed from zearalenone compared to β-zearalenol in humans. β-Zearalenol is about the same or slightly less potent as an estrogen relative to zearalenone.
 W
WZearalenone (ZEN), also known as RAL and F-2 mycotoxin, is a potent estrogenic metabolite produced by some Fusarium and Gibberella species. Particularly, is produced by Fusarium graminearum, Fusarium culmorum, Fusarium cerealis, Fusarium equiseti, Fusarium verticillioides, and Fusarium incarnatum.
 W
WZeranol, or zearanol, also known as α-zearalanol or simply zearalanol, is a synthetic nonsteroidal estrogen of the resorcylic acid lactone group related to mycoestrogens found in fungi in the Fusarium genus and is used mainly as an anabolic agent in veterinary medicine.