 W
WThermodynamics is expressed by a mathematical framework of thermodynamic equations which relate various thermodynamic quantities and physical properties measured in a laboratory or production process. Thermodynamics is based on a fundamental set of postulates, that became the laws of thermodynamics.
 W
WIn thermodynamics, Bridgman's thermodynamic equations are a basic set of thermodynamic equations, derived using a method of generating multiple thermodynamic identities involving a number of thermodynamic quantities. The equations are named after the American physicist Percy Williams Bridgman..
 W
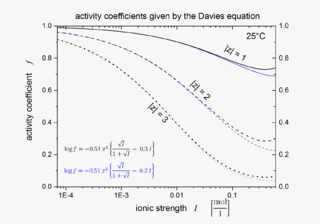
WThe Davies equation is an empirical extension of Debye–Hückel theory which can be used to calculate activity coefficients of electrolyte solutions at relatively high concentrations at 25 °C. The equation, originally published in 1938, was refined by fitting to experimental data. The final form of the equation gives the mean molal activity coefficient of an electrolyte that dissociates into ions having charges z1 and z2 as a function of ionic strength I:
 W
WThe Eötvös rule, named after the Hungarian physicist Loránd (Roland) Eötvös (1848–1919) enables the prediction of the surface tension of an arbitrary liquid pure substance at all temperatures. The density, molar mass and the critical temperature of the liquid have to be known. At the critical point the surface tension is zero.
 W
WIn thermodynamics, the fundamental thermodynamic relation are four fundamental equations which demonstrate how four important thermodynamic quantities depend on variables that can be controlled and measured experimentally. Thus, they are essentially equations of state, and using the fundamental equations, experimental data can be used to determine sought-after quantities like G or H. The relation is generally expressed as a microscopic change in internal energy in terms of microscopic changes in entropy, and volume for a closed system in thermal equilibrium in the following way.
 W
WMaxwell's relations are a set of equations in thermodynamics which are derivable from the symmetry of second derivatives and from the definitions of the thermodynamic potentials. These relations are named for the nineteenth-century physicist James Clerk Maxwell.
 W
WThis article is a summary of common equations and quantities in thermodynamics. SI units are used for absolute temperature, not Celsius or Fahrenheit.
 W
WA thermodynamic potential is a scalar quantity used to represent the thermodynamic state of a system. The concept of thermodynamic potentials was introduced by Pierre Duhem in 1886. Josiah Willard Gibbs in his papers used the term fundamental functions. One main thermodynamic potential that has a physical interpretation is the internal energy U. It is the energy of configuration of a given system of conservative forces and only has meaning with respect to a defined set of references. Expressions for all other thermodynamic energy potentials are derivable via Legendre transforms from an expression for U. In thermodynamics, external forces, such as gravity, are typically disregarded when formulating expressions for potentials. For example, while all the working fluid in a steam engine may have higher energy due to gravity while sitting on top of Mount Everest than it would at the bottom of the Mariana Trench, the gravitational potential energy term in the formula for the internal energy would usually be ignored because changes in gravitational potential within the engine during operation would be negligible. In a large system under even homogeneous external force, like the earth atmosphere under gravity, the intensive parameters should be studied locally having even in equilibrium different values in different places far from each other (see thermodynamic models of troposphere].