 W
WA desiccant is a hygroscopic substance that is used to induce or sustain a state of dryness (desiccation) in its vicinity; it is the opposite of a humectant. Commonly encountered pre-packaged desiccants are solids that absorb water. Desiccants for specialized purposes may be in forms other than solid, and may work through other principles, such as chemical bonding of water molecules. They are commonly encountered in foods to retain crispness. Industrially, desiccants are widely used to control the level of water in gas streams.
 W
WActivated alumina is manufactured from aluminium hydroxide by dehydroxylating it in a way that produces a highly porous material; this material can have a surface area significantly over 200 m²/g. The compound is used as a desiccant (to keep things dry by absorbing water from the air) and as a filter of fluoride, arsenic and selenium in drinking water. It is made of aluminium oxide (alumina; Al2O3). It has a very high surface-area-to-weight ratio, due to the many "tunnel like" pores that it has. Activated alumina in its phase composition can be represented only by metastable forms (gamma-Al2O3 etc.). Corundum (alpha-Al2O3), the only stable form of aluminum oxide, does not have such a chemically active surface and is not used as a sorbent.
 W
WCalcium carbide, also known as calcium acetylide, is a chemical compound with the chemical formula of CaC2. Its main use industrially is in the production of acetylene and calcium cyanamide.
 W
WCalcium chloride is an inorganic compound, a salt with the chemical formula CaCl2. It is a white coloured crystalline solid at room temperature, and it is highly soluble in water. It can be created by neutralising hydrochloric acid with calcium hydroxide.
 W
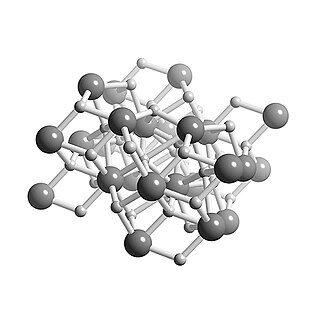
WCalcium hydride is the chemical compound with the formula CaH2, and is therefore an alkaline earth hydride. This grey powder (white if pure, which is rare) reacts vigorously with water liberating hydrogen gas. CaH2 is thus used as a drying agent, i.e. a desiccant.
 W
WCalcium oxide (CaO), commonly known as quicklime or burnt lime, is a widely used chemical compound. It is a white, caustic, alkaline, crystalline solid at room temperature. The broadly used term "lime" connotes calcium-containing inorganic materials, in which carbonates, oxides and hydroxides of calcium, silicon, magnesium, aluminium, and iron predominate. By contrast, quicklime specifically applies to the single chemical compound calcium oxide. Calcium oxide that survives processing without reacting in building products such as cement is called free lime.
 W
WCalcium sulfate (or calcium sulphate) is the inorganic compound with the formula CaSO4 and related hydrates. In the form of γ-anhydrite (the anhydrous form), it is used as a desiccant. One particular hydrate is better known as plaster of Paris, and another occurs naturally as the mineral gypsum. It has many uses in industry. All forms are white solids that are poorly soluble in water. Calcium sulfate causes permanent hardness in water.
 W
WCopper(II) sulfate, also known as copper sulphate, are the inorganic compounds with the chemical formula CuSO4(H2O)x, where x can range from 0 to 5. The pentahydrate (x = 5) is the most common form. Older names for this compound include blue vitriol, bluestone, vitriol of copper, and Roman vitriol.
 W
WThe Cromer cycle is a thermodynamic cycle that uses a desiccant to interact with higher relative humidity air leaving a cold surface. When a system is taken through a series of different states and finally returned to its initial state, a thermodynamic cycle is said to have occurred. The desiccant absorbs moisture from the air leaving the cold surface, releasing heat and drying the air, which can be used in a process requiring dry air. The desiccant is then dried by an air stream at a lower relative humidity, where the desiccant gives up its moisture by evaporation, increasing the air's relative humidity and cooling it. This cooler, moister air can then be presented to the same cold surface as above to take it below its dew point and dry it further, or it can be expunged from the system.
 W
WDiquat is the ISO common name for an organic dication that, as a salt with counterions such as bromide or chloride is used as a contact herbicide that produces desiccation and defoliation. Diquat is no longer approved for use in the European Union, although its registration in many other countries including the USA is still valid.
 W
WCalcium sulfate (or calcium sulphate) is the inorganic compound with the formula CaSO4 and related hydrates. In the form of γ-anhydrite (the anhydrous form), it is used as a desiccant. One particular hydrate is better known as plaster of Paris, and another occurs naturally as the mineral gypsum. It has many uses in industry. All forms are white solids that are poorly soluble in water. Calcium sulfate causes permanent hardness in water.
 W
WLithium chloride is a chemical compound with the formula LiCl. The salt is a typical ionic compound (with certain covalent characters), although the small size of the Li+ ion gives rise to properties not seen for other alkali metal chlorides, such as extraordinary solubility in polar solvents (83.05 g/100 mL of water at 20 °C) and its hygroscopic properties.
 W
WA lye is a metal hydroxide traditionally obtained by leaching wood ashes, or a strong alkali which is highly soluble in water producing caustic basic solutions. "Lye" most commonly refers to sodium hydroxide (NaOH), but historically has been used for potassium hydroxide (KOH).
 W
WMagnesium sulfate or magnesium sulphate is a chemical compound, a salt with the formula MgSO4, consisting of magnesium cations Mg2+ and sulfate anions SO2−4. It is a white crystalline solid, soluble in water but not in ethanol.
 W
WA molecular sieve is a material with pores of uniform size. These pore diameters are similar in size to small molecules, and thus large molecules cannot enter or be adsorbed, while smaller molecules can. As a mixture of molecules migrate through the stationary bed of porous, semi-solid substance referred to as a sieve, the components of highest molecular weight leave the bed first, followed by successively smaller molecules. Some molecular sieves are used in size-exclusion chromatography, a separation technique that sorts molecules based on their size. Other molecular sieves are used as desiccants.
 W
WMontmorillonite is a very soft phyllosilicate group of minerals that form when they precipitate from water solution as microscopic crystals, known as clay. It is named after Montmorillon in France. Montmorillonite, a member of the smectite group, is a 2:1 clay, meaning that it has two tetrahedral sheets of silica sandwiching a central octahedral sheet of alumina. The particles are plate-shaped with an average diameter around 1 μm and a thickness of 0.96 nm; magnification of about 25,000 times, using an electron microscope, is required to "see" individual clay particles. Members of this group include, amongst others, saponite, nontronite, beidellite, and hectorite.
 W
WSodium–potassium alloy, colloquially called NaK, is an alloy of the alkali metals sodium and potassium that is normally liquid at room temperature. Various commercial grades are available. NaK is highly reactive with water and may catch fire when exposed to air, so must be handled with special precautions.
 W
WNatron is a naturally occurring mixture of sodium carbonate decahydrate (Na2CO3·10H2O, a kind of soda ash) and around 17% sodium bicarbonate (also called baking soda, NaHCO3) along with small quantities of sodium chloride and sodium sulfate. Natron is white to colourless when pure, varying to gray or yellow with impurities. Natron deposits are sometimes found in saline lake beds which arose in arid environments. Throughout history natron has had many practical applications that continue today in the wide range of modern uses of its constituent mineral components.
 W
WPotassium is a chemical element with the symbol K and atomic number 19. Potassium is a silvery-white metal that is soft enough to be cut with a knife with little force. Potassium metal reacts rapidly with atmospheric oxygen to form flaky white potassium peroxide in only seconds of exposure. It was first isolated from potash, the ashes of plants, from which its name derives. In the periodic table, potassium is one of the alkali metals, all of which have a single valence electron in the outer electron shell, that is easily removed to create an ion with a positive charge – a cation, that combines with anions to form salts. Potassium in nature occurs only in ionic salts. Elemental potassium reacts vigorously with water, generating sufficient heat to ignite hydrogen emitted in the reaction, and burning with a lilac-colored flame. It is found dissolved in sea water, and occurs in many minerals such as orthoclase, a common constituent of granites and other igneous rocks.
 W
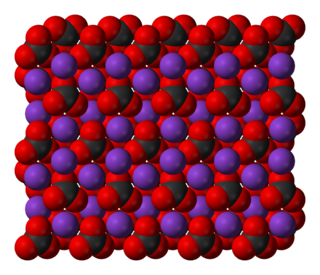
WPotassium carbonate is the inorganic compound with the formula K2CO3. It is a white salt, which is soluble in water. It is deliquescent, often appearing as a damp or wet solid. Potassium carbonate is mainly used in the production of soap and glass.
 W
WPotassium hydroxide is an inorganic compound with the formula KOH, and is commonly called caustic potash.
 W
WSilica gel is an amorphous and porous form of silicon dioxide (silica), consisting of an irregular tridimensional framework of alternating silicon and oxygen atoms with nanometer-scale voids and pores. The voids may contain water or some other liquids, or may be filled by gas or vacuum. In the last case, the material is properly called silica xerogel.
 W
WSodium is a chemical element with the symbol Na and atomic number 11. It is a soft, silvery-white, highly reactive metal. Sodium is an alkali metal, being in group 1 of the periodic table. Its only stable isotope is 23Na. The free metal does not occur in nature, and must be prepared from compounds. Sodium is the sixth most abundant element in the Earth's crust and exists in numerous minerals such as feldspars, sodalite, and rock salt (NaCl). Many salts of sodium are highly water-soluble: sodium ions have been leached by the action of water from the Earth's minerals over eons, and thus sodium and chlorine are the most common dissolved elements by weight in the oceans.
 W
WSodium chlorate is an inorganic compound with the chemical formula NaClO3. It is a white crystalline powder that is readily soluble in water. It is hygroscopic. It decomposes above 300 °C to release oxygen and leaves sodium chloride. Several hundred million tons are produced annually, mainly for applications in bleaching pulp to produce high brightness paper.
 W
WSodium hydroxide, also known as lye and caustic soda, is an inorganic compound with the formula NaOH. It is a white solid ionic compound consisting of sodium cations Na+ and hydroxide anions OH−.
 W
WSodium sulfate (also known as sodium sulphate or sulfate of soda) is the inorganic compound with formula Na2SO4 as well as several related hydrates. All forms are white solids that are highly soluble in water. With an annual production of 6 million tonnes, the decahydrate is a major commodity chemical product. It is mainly used as a filler in the manufacture of powdered home laundry detergents and in the Kraft process of paper pulping for making highly alkaline sulfides.
 W
WA sorbent is a material used to absorb or adsorb liquids or gases. Examples include:A material similar to molecular sieve material, which acts by adsorption. It has a large internal surface area and good thermal conductivity. It is typically supplied in pellets of 1 mm to 2 mm diameter and roughly 5 mm length or as grains of the order 1 mm. Occasionally as beads up to 5 mm diameter. They are typically made from aluminium oxide with a porous structure. Materials used to absorb other materials due to their high affinity for doing so. Examples include: In composting, dry materials absorb many odoriferous chemicals, and these chemicals help to decompose these sorbents. A sponge absorbs many times its own weight in water. A polypropylene fiber mat may be used to absorb oil. A cellulose fiber product may be used to absorb oil. The granular gel material in a baby diaper will absorb several times its original weight in urine.Desiccants attract water, drying out (desiccating) the surrounding materials.